Modelling and measuring intracellular competition for finite resources during gene expression
Dissecting the competition between genes for shared expressional resources is of fundamental importance for understanding the interplay between cellular components. Owing to the relationship between gene expression and cellular fitness, genomes are shaped by evolution to improve resource allocation. Whereas experimental approaches to investigate intracellular competition require technical resources and human expertise, computational models and in silico simulations allow vast numbers of experiments to be carried out and controlled easily, and with significantly reduced costs. Thus, modelling competition has a pivotal role in understanding the effects of competition on the biophysics of the cell. In this article, we review various computational models proposed to describe the different types of competition during gene expression. We also present relevant synthetic biology experiments and their biotechnological implications, and discuss the open questions in the field.

Architecture of Lipid Droplets in Endoplasmic Reticulum Is Determined by Phospholipid Intrinsic Curvature
Lipid droplets (LDs) store fats and play critical roles in lipid and energy homeostasis. They form between the leaflets of the endoplasmic reticulum (ER) membrane and consist of a neutral lipid core wrapped in a phospholipid monolayer with proteins. Two types of ER-LD architecture are thought to exist and be essential for LD functioning. Maturing LDs either emerge from the ER into the cytoplasm, remaining attached to the ER by a narrow membrane neck, or stay embedded in the ER and are surrounded by ER membrane. Here, we
identify a lipid-based mechanism that controls which of these two architectures is favored. Theoretical modeling indicated that the intrinsic molecular curvatures of ER phospholipids can determine whether LDs remain embedded in or emerge from the ER; lipids with negative intrinsic curvature such as diacylglycerol (DAG) and phosphatidylethanolamine favor LD embedding, while those with positive intrinsic curvature, like lysolipids, support LD emergence. This prediction was verified by altering the lipid composition of the ER in S. cerevisiae using mutants and the addition of exogenous lipids. We found that fat-storageinducing
transmembrane protein 2 (FIT2) homologs become enriched at sites of LD generation when biogenesis is induced. DAG accumulates at sites of LD biogenesis, and FIT2 proteins may promote LD emergence from the ER by reducing DAG levels at these sites. Altogether, our findings suggest that cells regulate LD integration in the ER by modulating ER lipid composition, particularly at sites of LD biogenesis and that FIT2 proteins may play a central role in this process.

Migrasome formation is mediated by assembly of micron-scale tetraspanin macrodomains
Migrasomes are recently discovered cellular organelles that form as large vesicle-like structures on retraction fibres of migrating cells. While the process of migrasome formation has been described before, the molecular mechanism underlying migrasome biogenesis remains unclear. Here, we propose that the mechanism of migrasome formation consists of the assembly of tetraspanin- and cholesterol-enriched membrane microdomains into micron-scale macrodomains, which swell into migrasomes. The major finding underlying the mechanism is that tetraspanins and cholesterol are necessary and sufficient for migrasome formation. We demonstrate the necessity of tetraspanins and cholesterol via live-cell experiments, and their sufficiency by generating migrasome-like structures in reconstituted membrane systems. We substantiate the mechanism by a theoretical model proposing that the key factor driving migrasome formation is the elevated membrane stiffness of the tetraspanin- and cholesterol-enriched macrodomains. Finally, the theoretical model was quantitatively validated by experimental demonstration of the membrane-stiffening effect of tetraspanin 4 and cholesterol.

Membrane Curvature and Tension Control the Formation and Collapse of Caveolar Superstructuresy title...
Caveolae, flask-shaped pits covered by caveolin-cavin coats, are abundant features of the plasma membrane of many cells. Besides appearing as single-membrane indentations, caveolae are organized as superstructures in the form of rosette-like clusters, whose mechanism of assembly and biological functions have been elusive. Here, we propose that clustering of caveolae in mature muscle cells is driven by forces originating from the elastic energy of membrane-bending deformations and membrane tension. We substantiate this mechanism by computational modeling, which recovers the unique shapes observed for the most ubiquitous caveolar clusters. We support the agreement between the calculated and observed configurations by electron tomography of caveolar clusters. The model predicts the experimentally assessable dependence of caveolar clustering on membrane tension and on the degree of the caveolar coat assembly. We reveal a difference in conformation and, possibly, in function and formation mechanism between caveolar clusters of muscle cells and of adipocytes.

Simultaneous detection of multiple DNA damage types by multicolour fluorescent labelling
Hereinwepresentanassayallowingconcurrentdetectionofoxidative DNA damage and photoproducts. We apply DNA repair enzymes specific for each lesion type to incorporate spectrally distinct fluorescent nucleotides, enabling simultaneous quantification of the lesions on individual DNA molecules. We follow the repair of both damage types in skin cells exposed to artificialsunlight.

Global modulation in DNA epigenetics during pro-inflammatory macrophage activation
DNA methylation patterns create distinct gene-expression profiles. These patterns are maintained after cell division, thus enabling the differentiation and maintenance of multiple cell types from the same genome sequence. The advantage of this mechanism for transcriptional control is that chemical-encoding allows to rapidly establish new epigenetic patterns ‘on-demand’ through enzymatic methylation and demethylation of DNA. Here we show that this feature is associated with the fast response of macrophages during their pro-inflammatory activation. By using a combination of mass spectroscopy and single-molecule imaging to quantify global epigenetic changes in the genomes of primary macrophages, we followed three distinct DNA marks (methylated, hydroxymethylated and unmethylated), involved in establishing new DNA methylation patterns during pro-inflammatory activation. The observed epigenetic modulation together with gene-expression data generated for the involved enzymatic machinery may suggest that de-methylation upon LPS-activation starts with oxidation of methylated CpGs, followed by excision-repair of these oxidized bases and their replacement with unmodified cytosine.

5‐Hydroxymethylcytosine as a clinical biomarker: Fluorescence‐based assay for high‐throughput epigenetic quantification in human tissues
Epigenetic transformations may provide early indicators for cancer and other disease. Specifically, the amount of genomic 5‐hydroxymethylcytosine (5‐hmC) was shown to be globally reduced in a wide range of cancers. The integration of this global biomarker into diagnostic workflows is hampered by the limitations of current 5‐hmC quantification methods. Here we present and validate a fluorescence‐based platform for high‐throughput and cost‐effective quantification of global genomic 5‐hmC levels. We utilized the assay to characterize cancerous tissues based on their 5‐hmC content, and observed a pronounced reduction in 5‐hmC level in various cancer types. We present data for glioblastoma, colorectal cancer, multiple myeloma, chronic lymphocytic leukemia and pancreatic cancer, compared to corresponding controls. Potentially, the technique could also be used to follow response to treatment for personalized treatment selection. We present initial proof‐of‐concept data for treatment of familial adenomatous polyposis.

Single Fluorescent Peptide Nanodots
Fluorescent peptide nanodots (PNDs) are bioorganic nanoparticles self-assembled from peptide biomolecules of different origin and complexity. These recently discovered nanodots of biological origin are highly promising for biomedical imaging applications due to their biocompatibility, bright and tunable fluorescence over the entire visible range and photostability. Here we apply single-particle microscopy methods to study the photophysical properties of individual PNDs. We show that the fluorescence spectrum tunability, studied previously only for PND ensembles in solutions, origins at the single-particle level. Temporal dynamics measurements of the single particles reveal fluorescence lifetime in the range of nanoseconds and pronounced fluorescence blinking with continuous bright states of seconds. The latter provides a first evidence of quantum emitter transitions between two states (ON and OFF) in fluorescent PNDs. All these findings advance the understanding of the fluorescence mechanism of PNDs and provide strong motivation for using PNDs as fluorescent agents for various bioimaging and super-resolution techniques.

Enzyme-free optical DNA mapping of the human genome using competitive binding
Optical DNA mapping (ODM) allows visualization of long-range sequence information along single DNA molecules. The data can for example be used for detecting long range structural variations, for aiding DNA sequence assembly of complex genomes and for mapping epigenetic marks and DNA damage across the genome. ODM traditionally utilizes sequence specific marks based on nicking enzymes, combined with a DNA stain, YOYO-1, for detection of the DNA contour. Here we use a competitive binding approach, based on YOYO-1 and netropsin, which highlights the contour of the DNA molecules, while simultaneously creating a continuous sequence specific pattern, based on the AT/GC variation along the detected molecule. We demonstrate and validate competitive-binding-based ODM using bacterial artificial chromosomes (BACs) derived from the human genome and then turn to DNA extracted from white blood cells. We generalize our findings with in-silico simulations that show that we can map a vast majority of the human genome. Finally, we demonstrate the possibility of combining competitive binding with enzymatic labeling by mapping DNA damage sites induced by the cytotoxic drug etoposide to the human genome. Overall, we demonstrate that competitive-binding-based ODM has the potential to be used both as a standalone assay for studies of the human genome, as well as in combination with enzymatic approaches, some of which are already commercialized.

SUN-LB009 Epigenetic Changes in Response to Metabolic Modifiers in Late Life: Exercise, High Fat Diet, and Angiotenin1-7 Effects on Metabolic Health and DNA Methylation in Frail Old Mice
Background: Reduced muscle and bone-mass and quality as well as impaired energy homeostasis are seen in both aging and the metabolic syndrome (MetS). We previously showed that exercise and Angiotensin1-7 (A1-7) ameliorate this burden in young rats, but their effect vis-a-vis aging is unknown. Aims: To assess the effects of exercise under normal and high-fat diet (HFD) with or without A1-7 on bone architecture and muscle quality as well as on musculoskeletal and fat epigenetic signatures in aged mice. Methods: 19-months-old-mice receiving HFD or normal-chow (NC) with or without 3-months exercise (treadmill, 6 days/week, 20 min/day or A1-7 (0.6 mcg/kg/d). Bone was evaluated by microCT. Global quantification of the epigenetic modification 5-methyl-or hydroxyl-methyl cytosine (5mC; 5hmc) by LC-MS/MS in white/brown adipose tissue (WAT; BAT) and Gastrocnemius muscle. Results: Old, sedentary mice were metabolically "starved" with low serum glucose and triglycerides. (A) Neither exercise nor high fat diet elicited sustained changes in body weight in old mice. B) Exercise per se rehabilitated glucose, triglycerides levels and muscle quality (enhanced muscle-fiber sectional area), accompanied by reduction in muscle 5mC and in BAT 5hmc (p<0.05). (C) Combination of exercise with A1-7 improved bone geometry and density in NC mice but not during HFD. Combination of A1-7 and exercise led to a reduction in global 5mC levels both in the gastrocnemius muscle and in WAT (p<0.05), thus suggesting that some of the effects maybe induced by changes in methylation patterns. Conclusions: The beneficial effects of A1-7 and exercise in aging skeletal tissue are operative only in NC but not in the presence of HFD. These beneficial effects are accompanied, and indeed, may be partly mediated by reversal of gene silencing as depicted by lesser DNA methylation in skeletal muscle. Finally, this is the first report that exercise can induce epigenetic changes in BAT, independent of changes in body weight. Unless otherwise noted, all abstracts presented at ENDO are embargoed until the date and time of presentation. For oral presentations, the abstracts are embargoed until the session begins. Abstracts presented at a news conference are embargoed until the date and time of the news conference. The Endocrine Society reserves the right to lift the embargo on specific abstracts that are selected for promotion prior to or during ENDO.

Long-read single-molecule maps of the functional methylome
We report on the development of a methylation analysis workflow for optical detection of fluorescent methylation profiles along chromosomal DNA molecules. In combination with BioNano Genomics genome mapping technology, these profiles provide a hybrid genetic/epigenetic genome-wide map composed of DNA molecules spanning hundreds of kilobase pairs (kbp). The method provides kbp-scale genomic methylation patterns comparable to whole genome bisulfite sequencing (WGBS) along genes and regulatory elements. These long single-molecule reads allow for methylation variation calling and analysis of large structural aberrations such as pathogenic macrosatellite arrays not accessible to single-cell second-generation sequencing. The method is applied here to study facioscapulohumeral muscular dystrophy (FSHD), simultaneously recording the haplotype, copy number and methylation status of the disease-associated, highly repetitive locus on Chromosome 4q.

Analytical epigenetics: single-molecule optical detection of DNA and histone modifications
The field of epigenetics describes the relationship between genotype and phenotype, by regulating gene expression without changing the canonical base sequence of DNA. It deals with molecular genomic information that is encoded by a rich repertoire of chemical modifications and molecular interactions. This regulation involves DNA, RNA and proteins that are enzymatically tagged with small molecular groups that alter their physical and chemical properties. It is now clear that epigenetic alterations are involved in development and disease, and thus, are the focus of intensive research. The ability to record epigenetic changes and quantify them in rare medical samples is critical for next generation diagnostics. Optical detection offers the ultimate single-molecule sensitivity and the potential for spectral multiplexing. Here we review recent progress in ultrasensitive optical detection of DNA and histone modifications.

Inferring broken detailed balance in the absence of observable currents
Identifying dissipation is essential for understanding the physical mechanisms underlying nonequilibrium processes. In living systems, for example, the dissipation is directly related to the hydrolysis of fuel molecules such as adenosine triphosphate (ATP). Nevertheless, detecting broken time-reversal symmetry, which is the hallmark of dissipative processes, remains a challenge in the absence of observable directed motion, flows, or fluxes. Furthermore, quantifying the entropy production in a complex system requires detailed information about its dynamics and internal degrees of freedom. Here we introduce a novel approach to detect time irreversibility and estimate the entropy production from time-series measurements, even in the absence of observable currents. We apply our technique to two different physical systems, namely, a partially hidden network and a molecular motor. Our method does not require complete information about the system dynamics and thus provides a new tool for studying nonequilibrium phenomena.

Shape-Dependent Interactions of Manganese Oxide Nanomaterials with Lipid Bilayer Vesicles
Interactions of transition-metal-oxide nanomaterials with biological membranes have important environmental implications and applications in ecotoxicity and life-cycle assessment analysis. In this study, we quantitatively assess the impact of MnO2 nanomaterial morphology—1-dimentional (1D) nanowires, 2D nanosheets, and 3D nanoflowers—on their interaction with phospholipid vesicles as a model for biological membranes. Confocal microscopy suggests visual evidence for the interaction of undisrupted vesicles with dispersed MnO2 nanomaterials of different morphologies, and it further supports the observation that minimal dye leakage of the vesicle inner solution was detected during the interaction with MnO2 nanomaterials during the dye leakage assay. Upon titration of vesicles to dispersions of MnO2 nanowires, nanosheets, and nanoflowers, each roughly 10 times larger than the vesicles, dynamic light scattering reveals two diffusive time scales associated with aggregates in the mixture. While the longer time scale corresponds to the dispersed MnO2 control population, the appearance of a shorter timescale with vesicle addition indicates interaction between the dispersed metal oxide nanomaterials and the vesicles. The interaction is shape-dependent, being more pronounced for MnO2 nanowires than for nanosheets and nanoflowers. Furthermore, the shorter diffusive time scale is intermediate between the vesicle and nanomaterial controls, which may suggest a degree of metal oxide aggregate breakup. Vesicle adsorption isotherms and zeta potential measurements during titration corroborate vesicle attachment on the nanomaterials. Our results suggest that dispersed nanomaterial shape plays an important role in mediating non-destructive vesicle-nanomaterial interactions and that lipid vesicles act as efficient surfactants for MnO2 nanomaterials.

Nonequilibrium associative retrieval of multiple stored self-assembly targets
Major experimental research efforts have gone into investigating general principles governing self-assembly under nonequilibrium driving. However, in contrast to equilibrium scenarios, where the system tends to find local minima in the free-energy landscape, there is no equivalent theoretical framework for systems operating far from equilibrium. Inspired by many examples of nonequilibrium self-assembly in living systems, we set out to explore the added benefits achieved by nonequilibrium driving and identify distinctive collective phenomena that emerge in this regime. We demonstrate the interplay between the assembly speed, kinetic stability, and relative population of dynamical attractors, aiming to provide insights into nonequilibrium self-assembly processes and lay the foundations for understanding biomolecular cases as well as for designing examples.

Charge regulation with fixed and mobile charged macromolecules
Uncompensated charges do not usually occur in Nature and any local charge should be a result of charge separation. Dissociable chemical groups at interfaces in contact with ions in solution, whose chemical equilibrium depends both on short-range non-electrostatic and long-range electrostatic interactions, are the physical basis of this charge separation, known as charge regulation phenomena. The charged groups can be either fixed and immobile, as in the case of solvent exposed solid substrate and soft bounding surfaces (e.g., atomically smooth mica surfaces and soft phospholipid membranes), or free and mobile, as in the case of charged macro-ions (e.g., protein or other biomolecules). Here, we review the mean-field formalism used to describe both cases, with a focus on recent advances in the modeling of mobile charge-regulated macro-ions in an ionic solution. The general form of the screening length is derived, and is shown to combine the concept of intrinsic capacitance (introduced by Lund and Jönsson) with bulk capacitance, resulting from the mobility of small ions and macro-ions. The advantages and disadvantages of different formulations, such as the cell model vs. the collective approach, are discussed, along with several suggestions for future experiments and modeling.

Charge regulating macro-ions in salt solutions: screening properties and electrostatic interactions
We revisit the charge-regulation mechanism of macro-ions and apply it to mobile macro-ions in a bathing salt solution. In particular, we examine the effects of correlation between various adsorption/desorption sites and analyze the collective behavior in terms of the solution effective screening properties. We show that such a behavior can be quantified in terms of the charge asymmetry of the macro-ions, defined by their preference for a non-zero effective charge, and their donor/acceptor propensity for exchanging salt ions with the bathing solution. Asymmetric macro-ions tend to increase
the screening, while symmetric macro-ions can in some cases decrease it. Macro-ions that are classified as donors display a rather regular behavior, while those that behave as acceptors exhibit an anomalous non-monotonic Debye length. The screening properties, in their turn, engender important modifications to the disjoining pressure between two charged surfaces. Our findings are in particular relevant for solutions of proteins, whose exposed amino acids can undergo charge dissociation/association processes to/from the bathing solution, and can be considered as a solution of charged regulated macro-ions, as analyzed here.

Broad spectrum detection of DNA damage by Repair Assisted Damage Detection (RADD).
Environmental exposures, reactive by-products of cellular metabolism, and spontaneous deamination events result in a spectrum of DNA adducts that if un-repaired threaten genomic integrity by inducing mutations, increasing instability, and contributing to the initiation and progression of cancer. Assessment of DNA adducts in cells and tissues is critical for genotoxic and carcinogenic evaluation of chemical exposure and may provide insight into the etiology of cancer. Numerous methods to characterize the formation of DNA adducts and their retention for risk assessment have been developed. However, there are still significant drawbacks to the implementation and wide-spread use of these methods, because they often require a substantial amount of biological sample, highly specialized expertise and equipment, and depending on technique, may be limited to the detection and quantification of only a handful of DNA adducts at a time. There is a pressing need for high throughput, easy to implement assays that can assess a broad spectrum of DNA lesions, allowing for faster evaluation of chemical exposures and assessment of the retention of adducts in biological samples. Here, we describe a new methodology, Repair Assisted Damage Detection (RADD), which utilizes a DNA damage processing repair enzyme cocktail to detect and modify sites of DNA damage for a subsequent gap filling reaction that labels the DNA damage sites. This ability to detect and label a broad spectrum of DNA lesions within cells, offers a novel and easy to use tool for assessing levels of DNA damage in cells that have been exposed to environmental agents or have natural variations in DNA repair capacity.

Selective nanopore sequencing of human BRCA1 by Cas9-assisted targeting of chromosome segments (CATCH)
Next generation sequencing (NGS) is challenged by structural and copy number variations larger than the typical read length of several hundred bases. Third-generation sequencing platforms such as single-molecule real-time (SMRT) and nanopore sequencing provide longer reads and are able to characterize variations that are undetected in NGS data. Nevertheless, these technologies suffer from inherent low throughput which prohibits deep sequencing at reasonable cost without target enrichment. Here, we optimized Cas9-Assisted Targeting of CHromosome segments (CATCH) for nanopore sequencing of the breast cancer gene BRCA1. A 200 kb target containing the 80 kb BRCA1 gene body and its flanking regions was isolated intact from primary human peripheral blood cells, allowing long-range amplification and long-read nanopore sequencing. The target was enriched 237-fold and sequenced at up to 70× coverage on a single flow-cell. Overall performance and single-nucleotide polymorphism (SNP) calling were directly compared to Illumina sequencing of the same enriched sample, highlighting the benefits of CATCH for targeted sequencing. The CATCH enrichment scheme only requires knowledge of the target flanking sequence for Cas9 cleavage while providing contiguous data across both coding and non-coding sequence and holds promise for characterization of complex disease-related or highly variable genomic regions.

Epigenetic Optical Mapping of 5-Hydroxymethylcytosine in Nanochannel Arrays
The epigenetic mark 5-hydroxymethylcytosine (5-hmC) is a distinct product of active DNA demethylation that is linked to gene regulation, development, and disease. In particular, 5-hmC levels dramatically decline in many cancers, potentially serving as an epigenetic biomarker. The noise associated with next-generation 5-hmC sequencing hinders reliable analysis of low 5-hmC containing tissues such as blood and malignant tumors. Additionally, genome-wide 5-hmC profiles generated by short-read sequencing are limited in providing long-range epigenetic information relevant to highly variable genomic regions, such as the 3.7 Mbp disease-related Human Leukocyte Antigen (HLA) region. We present a long-read, highly sensitive single-molecule mapping technology that generates hybrid genetic/epigenetic profiles of native chromosomal DNA. The genome-wide distribution of 5-hmC in human peripheral blood cells correlates well with 5-hmC DNA immunoprecipitation (hMeDIP) sequencing. However, the long single-molecule read-length of 100 kbp to 1 Mbp produces 5-hmC profiles across variable genomic regions that failed to show up in the sequencing data. In addition, optical 5-hmC mapping shows a strong correlation between the 5-hmC density in gene bodies and the corresponding level of gene expression. The single-molecule concept provides information on the distribution and coexistence of 5-hmC signals at multiple genomic loci on the same genomic DNA molecule, revealing long-range correlations and cell-to-cell epigenetic variation.

Hypersensitive quantification of global 5-hydroxymethylcytosine by chemoenzymatic tagging
5-hydroxymethylcytosine (5hmC) is an epigenetic DNA modification. Tissue-specific reduction in global 5hmC levels has been associated with various types of cancer. One of the challenges associated with detecting 5hmC levels is its extremely low content, especially in blood. The gold-standard for reliable global 5hmC quantitation is liquid chromatography-tandem mass spectroscopy (LC-MS/MS) operating in a multiple reaction monitoring (MRM) mode. Difficulties associated with 5hmC detection by LC-MS/MS include its low abundance, low ionization efficiency and possible ion suppression from co-eluted compounds. Hence, detecting 5hmC levels in blood samples for diagnosis of leukemia and other blood malignancies presents a unique challenge. To overcome these difficulties we introduce a simple chemoenzymatic method for specifically tagging 5hmC, resulting in an eight-fold increase in detection sensitivity. We demonstrate that we could quantitatively detect 5hmC levels in various human tissues, including blood samples from healthy individuals and leukemia patients, using the most basic quadrupole mass-analyzer instrument and only 200 ng of DNA. The limit of detection (LOD) of our technique is 0.001% 5hmC from 300 ng DNA, sufficient for future mass-spectroscopy based diagnostics of diseases associated with low 5hmC levels such as leukemia.

Localization of random walks to competing manifolds of distinct dimensions
We consider localization of a random walk (RW) when attracted or repelled by multiple extended manifolds of different dimensionalities. In particular, we focus on (d−1)- and (d−2)-dimensional manifolds in d-dimensional space, where attractive interactions are (fully or marginally) relevant. The RW can then be in one of four phases where it is localized to neither, one, or both manifolds. The four phases merge at a special multi-critical point where (away from the manifolds) the RW spreads diffusively. Extensive numerical analyses on two dimensional RWs confined inside or outside a rectangular wedge confirm general features expected from a continuum theory, but also exhibit unexpected attributes, such as a reentrant localization to the corner while repelled by it.
Pathological transitions in myelin membranes driven by environmental and multiple sclerosis conditions
In demyelination diseases, such as multiple sclerosis, the structure of the axons’ protective sheaths is disrupted. Due to the proximity of cytoplasmic myelin membrane to structural phase transition, minor alterations in the local environmental conditions can have devastating results. Using small-angle X-ray scattering and cryogenic transmission electron microscopy, we show that drastic structural reorganization and instabilities of myelin membrane are linked to specific environmental conditions and molecular composition in healthy and diseased states. These instabilities involve phase transition from the healthy lamellar membranes to pathological inverted hexagonal phase. These results highlight that local environmental conditions are critical for myelin function and should be considered as alternative routes for early pathology and as a means to avoid the initiation of demyelination.
Nonlinear Elasticity of the ECM Fibers Facilitates Efficient Intercellular Communication
The forces that cells apply against their surroundings are critical in many biological functions including migration and differentiation. In the current issue of Biophysical Journal (Oct 2018), Sopher et al. presented a computational study of two contractile cells embedded in fibrous networks, environment that mimic the extracellular matrix (ECM). They showed how a unique mechanical behavior of the ECM- a highly non-linear response to tensile and compressive forces- contributes to directional forces and deformations transmitted in the matrix between neighboring cells. This mechanism may allow cells to more efficiently interact over long distances, and can impact various biological processes.
Quantitative Analysis of Delta-like 1 Membrane Dynamics Elucidates the Role of Contact Geometry on Notch Signaling
Khait et al. show large cell-to-cell variability in the diffusion coefficients of the Notch ligand Delta-like 1. A combination of quantitative FRAP-TIRF imaging and mathematical modeling is used to examine the implications of this result on Notch signaling and its dependence on cell-cell contact geometry.
Cell-Cell Contact Area Affects Notch Signaling and Notch-Dependent Patterning
Cell geometry varies considerably between cells in almost every tissue.
Shaya et al. show that Notch signaling between pairs of cells correlates with their contact area. This dependence of signaling on contact area can bias Notchmediated cell fate decisions such as those occurring in chick inner ear development.

The atypical cadherins Fat and Dachsous (Ds) have been found to underlie planar cell polarity (PCP) in many tissues. Theoretical models suggest that polarity can arise from localized feedbacks on Fat-Ds complexes at the cell boundary. However, there is currently no direct
evidence for the existence or mechanism of such feedbacks. To directly test the localized feedback model, we developed a synthetic biology platform based on mammalian cells expressing the human Fat4 and Ds1. We show that Fat4-Ds1 complexes accumulate on cell boundaries in a
threshold-like manner and exhibit dramatically slower dynamics than unbound Fat4 and Ds1. This suggests a localized feedback mechanism based on enhanced stability of Fat4-Ds1 complexes. We also show that co-expression of Fat4 and Ds1 in the same cells is sufficient to induce polarization of Fat4-Ds1 complexes. Together, these results provide direct evidence that localized feedbacks on Fat4-Ds1 complexes can give rise to PCP.
DOI: https://doi.org/10.7554/eLife.24820.001

Electrostatics of patchy surfaces
In the study of colloidal, biological and electrochemical systems, it is customary to treat surfaces, macromolecules and electrodes as homogeneously charged. This simplified approach is proven successful in most cases, but fails to describe a wide range of heterogeneously charged surfaces commonly used in experiments. For example, recent experiments have revealed a long-range attraction between overall neutral surfaces, locally charged in a mosaic-like structure of positively and negatively charged domains (“patches”). Here, we review experimental and theoretical studies addressing the stability of heterogeneously charged surfaces, their effect on ionic profiles in solution, and the interaction between two such surfaces. We focus on electrostatics, and highlight the important new physical parameters appearing in the heterogeneous case, such as the largest patch size and inter-surface charge correlations.
Kinetically constrained models are lattice-gas models that are used for describing glassy systems. By construction, their equilibrium state is trivial and there are no equal-time correlations between the occupancy of different sites. We drive such models out of equilibrium by connecting them to two reservoirs of different densities, and we measure the response of the system to this perturbation.
We show how including attraction in interparticle interactions reverses the effect of fluctuations in ordering of a prototypical artificial frustrated system. Buckled colloidal monolayers exhibit the same ground state as the Ising antiferromagnet on a deformable triangular lattice, but it is unclear if ordering in the two systems is driven by the same geometric mechanism
The organization of live cells to tissues is associated with the mechanical interaction between cells, which is mediated through their elastic environment. We model cells as spherical active force dipoles surrounded by an infinite elastic matrix, and analytically evaluate the interaction energy for different scenarios of their regulatory behavior.
The structural complexity of metamaterials is limitless, but, in practice, most designs comprise periodic architectures that lead to materials with spatially homogeneous features. More advanced applications in soft robotics, prosthetics and wearable technology involve spatially textured mechanical functionality, which requires aperiodic architectures
The biological function of protein assemblies has been conventionally equated with a unique three-dimensional protein structure and protein-specific interactions. However, in the past 20 years it has been found that some assemblies contain long flexible regions that adopt multiple structural conformations. These include neurofilament proteins that constitute the stress-responsive supportive network of neurons. Herein, we show that the macroscopic properties of neurofilament networks are tuned by enzymatic regulation of the charge found on the flexible protein regions. The results reveal an enzymatic (phosphorylation) regulation of macroscopic properties such as orientation, stress response, and expansion in flexible protein assemblies. Using a model that explains the attractive electrostatic interactions induced by enzymatically added charges, we demonstrate that phosphorylation regulation is far richer and versatile than previously considered.
Abstract Evolutionary expansion of signaling pathway families often underlies the evolution of regulatory complexity. Expansion requires the acquisition of a novel homologous pathway and the diversification of pathway specificity. Acquisition can occur either vertically, by duplication, or through horizontal transfer, while divergence of specificity is thought to occur through a promiscuous protein intermediate.
In demyelinating diseases such as multiple sclerosis, disrupted myelin structures impair the functional role of the sheath as an insulating layer for proper nerve conduction. Though the etiology and recovery pathways remain unclear, in vivo studies show alterations in the lipid and the adhesive protein (myelin basic protein, MBP) composition. We find that in vitro cytoplasmic myelin membranes with modified lipid composition and low MBP concentration, as in demyelinating disease, show structural instabilities and pathological phase transition from a lamellar to inverted hexagonal, which involve enhanced local curvature. Similar curvatures are also found in vivo in diseased myelin sheaths.
Neurofilament proteins form a filamentous network that mechanically supports nerve cells, and they are critical for structural integrity and proper neuronal function. These proteins are often exposed to large compression stress, resulting, for example, from the transport of cellular organelles through the network. But it is not clear how neurofilament proteins can withstand such stresses. We have experimentally measured and theoretically modeled the compression response of neurofilament proteins, showing that these proteins function like shock absorbers.
Abstract Quorum sensing is a process of chemical communication that bacteria use to monitor cell density and coordinate cooperative behaviors. Quorum sensing relies on extracellular signal molecules and cognate receptor pairs.
We investigate a three-dimensional kinetically-constrained model that exhibits two types of phase transitions at different densities. At the jamming density there is a mixed-order phase transition in which a finite fraction of the particles become frozen, but the other particles may still diffuse throughout the system.
G protein-gated K+ channels (GIRK; Kir3), activated by Gβγ subunits derived from Gi/o proteins, regulate heartbeat and neuronal excitability and plasticity. Both neurotransmitter-evoked (Ievoked) and neurotransmitter-independent basal (Ibasal) GIRK activities are physiologically important, but mechanisms of Ibasal and its relation to Ievoked are unclear.
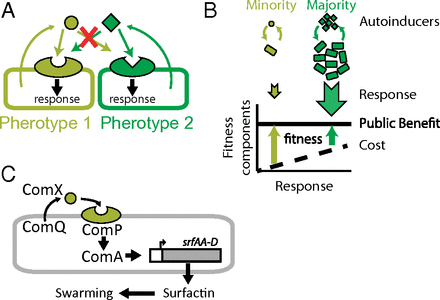
Abstract Bacterial quorum sensing enables bacteria to cooperate in a density-dependent manner via the group-wide secretion and detection of specific autoinducer molecules. Many bacterial species show high intraspecific diversity of autoinducer–receptor alleles, called pherotypes.
Metastable-to-stable phase transition is an uncontrolled spontaneous process, highly sensitive to microscopic details. It is often assumed that this transition occurs by overcoming an activation barrier through a stochastic process. However, for the case of self-assembled lipid suspensions that exhibit such phase-transition, the driving mechanisms and dynamics are not well understood. Using cryogenic TEM and solution x-ray scattering we studied the phase transition dynamics of supercooled lipid-based particles and found a unique metastable phase transition from liquid-crystalline to stable crystalline state. Unlike conventional supercooling phase transitions, upon cooling from 60°C to 37°C, recrystallization is delayed by tens of hours in a robust, predetermined and temporally controlled manner. Our results suggest that a non-stochastic physical mechanism is responsible for the delayed recrystallization, involving several rate-affecting processes.